Cyclic voltammetry has been widely used in electrochemical research due to its features of simple experiment, abundant information and theoretical analysis. For example, it can be used to quantitatively analyse the peak current, qualitatively analyse the peak potential difference, determine the reversibility of electrode processes, explore the electrochemical behaviour of unknown electrochemical systems, and apply it in various fields of applied electrochemistry.
The pseudocapacitance effect is a reversible Faraday charge transfer reaction that occurs at or near the surface of the material, and is a fast interfacial lithium storage behaviour. In cyclic voltammetry test, the cyclic voltammetry curve of such materials is similar to the rectangular shape of a capacitor, even if there is a redox peak, it is a wide bun peak, and the peak potential difference is small, so the reversibility is very good; in the process of constant-current charging and discharging, the charging and discharging curves of such materials is a tilted straight line, and the △Q / △E at any point of the straight line is almost equal, and the voltage hysteresis is very small, which is a typical electrochemical capacitor characteristics, so we call this kind of fast interfacial lithium storage behaviour. capacitor characteristics, so such a rapid interfacial energy storage reaction is called pseudocapacitance effect.
Conventional capacitors are mostly based on high specific surface area materials for the positive and negative electrodes, and the electrochemical reaction process is a typical adsorption and detachment mechanism. Activated carbon and other electrode materials with high specific surface area as the positive and negative electrodes can provide sufficient ionic adsorption and detachment sites. The electrochemical energy storage based on pseudocapacitance mechanism can greatly enhance the lithium storage capacity of the electrode materials by shifting the ion reaction to the surface or near surface of the materials. Therefore, pseudocapacitors based on pseudocapacitive energy storage mechanism have gradually replaced the traditional double layer capacitors and become the next generation of power energy storage devices.
In order to analyse the pseudocapacitive lithium storage behaviour of electrode materials. In order to analyse the pseudocapacitive lithium storage behaviour of the electrode material, a cyclic voltammetric curve based on the Nb2O5/Li half-cell can be taken for the determination.
As shown in Fig. 1, the experimental setup is composed of an electrochemical workstation and a Nb2O5/Li half-cell.
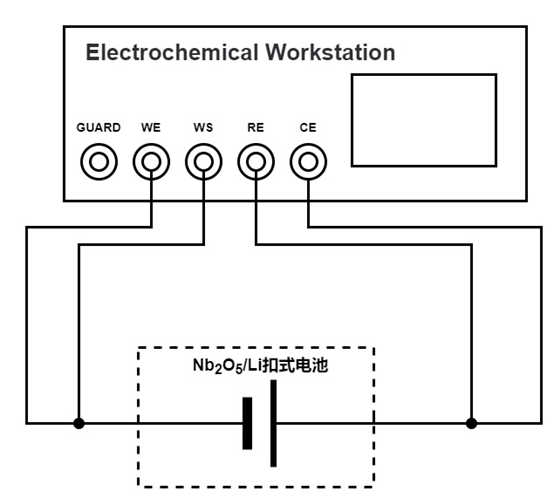
Fig. 1 Schematic diagram of the electrochemical workstation and the experimental setup for Nb2O5/Li half-cells
Take the processing of Nb2O5 electrode as an example, in order to calculate the pseudocapacitance CV curve under a certain sweep speed, we can take one point every 0.1 V (intercept 1.1 V.1.2V.1.3 V......3,0 V), and take 20 points for each of the positive and negative sweeps, and the more points we take, the smoother the pseudocapacitance CV curve is. Calculate the pseudo-capacitance current-break potential by potential, and then connect all points in series to get the pseudo-capacitance CV curve. Similarly, the simulated CV curve of pseudocapacitance current can be obtained by selecting the pseudocapacitance current with different potentials under the same sweeping speed, and the ratio of the area of the pseudocapacitance CV curve to the measured CV curve can be obtained by calculating the ratio of the CV curve to the area of the measured CV curve as shown in Fig. 2, which can be obtained by changing the lithium storage ratio contributed by pseudocapacitance under the sweeping speed.
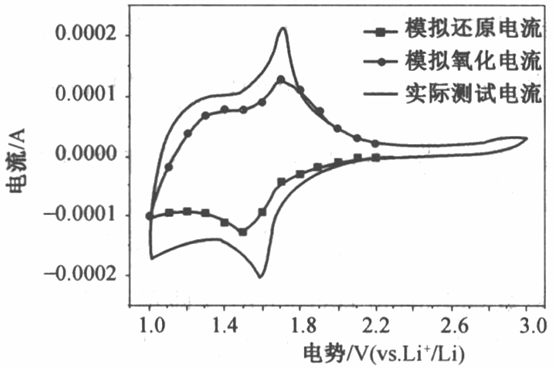
Fig. 2 CV curves of the current contribution of the simulated pseudocapacitor