1.Definition of Powder Porous Electrode
In the chemical power supply, rarely used by a single material manufactured plate electrode (or called the whole electrode), and most of the powder porous electrode. For example, the positive and negative electrodes of lead-acid batteries, zinc and silver batteries, zinc electrode and silver electrode. The so-called powder porous electrode, is composed of powder (mainly electrode reaction of the powdered active substance and other components) and the skeleton. Such electrodes can have a high porosity and specific surface, so that the actual working current density of the electrode is greatly reduced for the same apparent area, reducing the electrochemical polarization. In conclusion, the use of porous electrodes makes the performance of chemical power supply greatly improved, and provides a broad prospect for the development and use of various new electrodes.
Determining the true surface area of an electrode is an important task in electrochemical production and scientific research. Because the electrode reaction is concentrated on the electrode/solution interface. Therefore, for an electrode, when its chemical composition, structure, physical state, etc. are the same, its activity depends on the size of the surface area. The true surface area of a powder porous electrode can be hundreds or even tens of thousands of times the apparent area.
There are many methods to measure the true surface area, such as gas adsorption method, physical method and electrochemical method. In this experiment, the electrochemical method is used, that is, constant potential step method (or constant potential square wave method). The essence of the constant potential step method to measure the real surface area of the electrode is to determine the double layer capacitance of the electrode, and then calculate the real surface area of the electrode.
2. Constant potential step method
Constant potential step transient process is characterized by the start of the transient experiment before the polarization current is zero, the research electrode is in the open-circuit potential (equilibrium potential or stabilized potential), the beginning of the experiment, the research electrode potential suddenly jumped to a specified constant value until the end of the experiment, while recording the change rule of the polarization current with time. The step waveform imposed by the constant potential step method is shown in Figure 1.
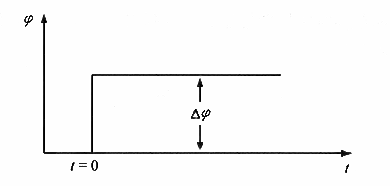
Fig. 1. Potential step signal applied in the constant potential step method
To the electrode at equilibrium potential or stable potential is suddenly added a small amplitude potential step signal, and the duration is not too long, so that the electrode potential in the equilibrium potential or stable potential fluctuations in the vicinity of the electrode potential, at this time it can be considered that the electrochemical reaction resistor Rct and the double layer capacitance Cd is a constant, the effect of concentration polarization can be ignored, so the equivalent circuit of the electrode is shown in Figure 2.
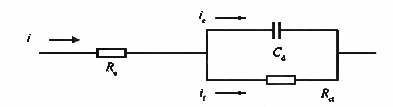
Fig. 2. Equivalent circuit of the electrode process in the absence of concentration polarization
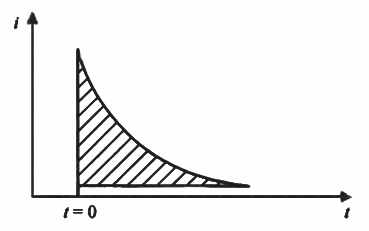
Obviously to determine the double layer capacitance of the electrode, conditions should be created so that the system is in the ideal polarized electrode, that is, in the controlled potential range, the electrode basically does not electrochemical reaction (Rct → ∞), and so the equivalent circuit is simplified from Fig. 2 to Fig. 3. When Rct → ∞, if → 0, at this time, the polarization current i is ic. Response waveforms are shown in Fig. 4. the magnitude of the ∆q is measured from Fig. 4, and is substituted into the following way, it can be Calculate the double layer capacitance Cd.
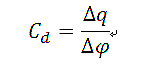
Fig. 3. Equivalent circuit in the absence of concentration polarization and electrochemical reaction
The double layer capacitance of an electrode is directly proportional to the true surface area of the electrode. Pure mercury has the smoothest surface, so it can be assumed that the apparent area of pure mercury is equal to its true surface area. The double layer capacitance of a mercury electrode is known to be 20μF-cm-2, which is taken as the standard and denoted as CN, which represents the capacitance per unit of real surface area. By dividing the measured capacitance value of the electrode, Cd, by CN, the total true surface area, S, of the electrode can be calculated.For example:

Experiments were carried out to measure the true surface area of silver sheet electrodes and silver powder pressed electrodes using electrochemical workstation SE1106. The electrolytic cell was a three-electrode system, the auxiliary electrode was a platinum sheet, the reference electrode was a mercuric oxide electrode, and the electrolyte was a 1 mol -L-1 KOH solution. The silver sheet and silver powder pressed electrode were used sequentially as the study electrodes. Timed current method was chosen (Fig. 5) starting potential was open circuit potential, high potential was open circuit potential +10mV, step time was 10s, and current time curve was recorded.
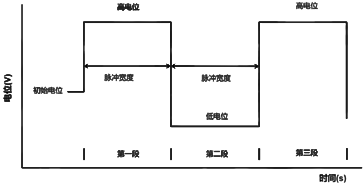
Fig 5. Timing current method