The precipitation of hydrogen is the most common reaction in the aqueous solution system, is the basic reaction of electrolysis of water, but also the secondary battery charging process, electroplating process is often accompanied by side reactions, therefore, is a common and important reaction in the electrochemical system.
1. Precipitation Pattern of Hydrogen on Metal Electrodes
Hydrogen precipitation on a metal electrode has the following pattern:
(1) On most metals, the H2 precipitation reaction has to take place at high ηc, i.e., in accordance with the Tafel equation
ηc = a + b *lg i
Where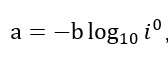
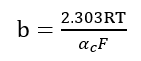 .
.
(2) Different systems (different electrodes or electrolytes) have different a and b values, see Table 1.
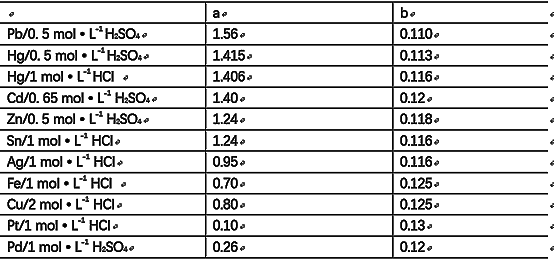
Table 1. a, b values in Tafel's formula for different systems
(3) The b-value has a small range of 0.11 to 0.13 (around 0.12).
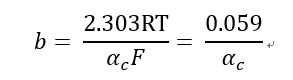
When T is certain, the value of b expresses the degree of activation energy influence at ηc. Similar values of b indicate that ηc influences the activation energy of the H precipitation process to approximately the same extent. Higher b values (>140 mV) are observed if the surface state has changed within the potential range involved.
(4) Different electrode systems, a value of the difference is large, for 0.1 ~ 1.5.
It is known that the larger the io, the better the reversibility of the reaction, so can also be used to judge or compare the value of a reversibility of good and bad; io larger, a smaller, the better the reversibility, but also the catalytic ability of the reaction is great, the electrode in the precipitation of H only requires a small overpotential; conversely, the smaller the io, the larger the a, the worse the reversibility of the reaction of the electrode catalytic ability of the electrode small, the electrode on the precipitation of H2 is often required to be a larger overpotential. Therefore, the commonly used electrode materials can be classified into three categories according to the size of a value:
High ηH2 metals: (a = 1.0 to 1.5) Pb, Cd, Hg, Zn , Sb, Bi, Sn, Tl.
Medium-ηH2 metals: (a=0.5-0.9) Fe, Co, Ni, Cu, Ag, Au.
Low ηH2 metals: (a = 0.1 to 0.3) Pt, Pd, Ru.
Where ηH2, is the hydrogen overpotential, which refers to the cathodic overpotential required for the reduction reaction of H2 precipitation. The hydrogen overpotential is usually tested by the linear potential scanning method.
An appropriate amount of 1 mol - L-1H2SO4 solution was selected as the electrolytic cell, Pt electrode was used as the working electrode, Pt electrode was selected as the auxiliary electrode, and the electrolytic cell was acidic, so the mercury sulphate reference electrode was selected. Linear Scanning Voltammetry (LSV) was selected for the experiment of hydrogen precipitation by electrolysis of water using an electrochemical workstation, where WS, WE were connected to the working electrode, RE was connected to the auxiliary electrode and CE was connected to the reference electrode.
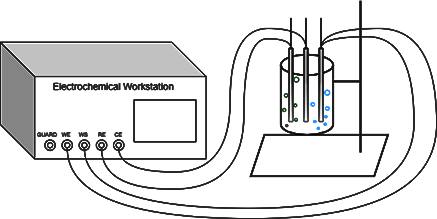
What occurs at the working electrode is an oxygen precipitation reaction at the anode, with bubbles (blue) being produced near the electrode, whereas what occurs at the auxiliary electrode is a hydrogen precipitation reaction at the cathode, which gradually intensifies as the cathodic potential grows, and produces bubbles (green) at a faster rate than at the working electrode.
2. Possible Reaction Mechanisms of Hydrogen Precipitation Process on Different Metals
Although the precipitation of hydrogen on metal has the above law, the reaction mechanism is not the same when precipitating on different metals.
There are three possible surface steps in the precipitation of hydrogen:
Electrochemical step: H+ + M + e- ⇌ MH
Complex desorption step: MH + MH ⇌ H2 x 2M
Electrochemical desorption step: MH + H+ + e- ⇌ H2 ten M
In high hydrogen overpotential metals, hydrogen precipitation proceeds by a delayed discharge mechanism, i.e., the electrochemical step is the control step of the reaction.
In the middle and low hydrogen overpotential metals, the mechanism of hydrogen precipitation is more complicated, and the mechanism of hydrogen precipitation may be a compound desorption mechanism or electrochemical desorption mechanism, or even the same metal in different polarisation conditions with different hydrogen precipitation mechanisms. Taking the Pt electrode as an example, due to the easy generation of adsorbed hydrogen atoms on the surface of Pt, it is usually controlled by compound desorption in the low polarisation region, while the high polarisation region is explained by electrochemical desorption mechanism.